Antimony is a metalloid element used mainly to harden lead alloys, boost flame retardant performance in plastics and textiles, and act as a catalyst or stabilizer in polymer production. Its practical value comes from three concrete properties: it strengthens grid plates in lead‑acid batteries for longer cycle life, it enhances halogenated flame retardant systems to reduce flammability, and certain antimony oxides speed PET synthesis as a catalyst.
Common pitfalls include uncontrolled particle contamination in recycled plastics and regulatory limits on specific antimony compounds that affect formulation choices. A surprising tradeoff many engineers overlook is how small changes in oxidation state alter both performance and environmental risk, shaping material selection.
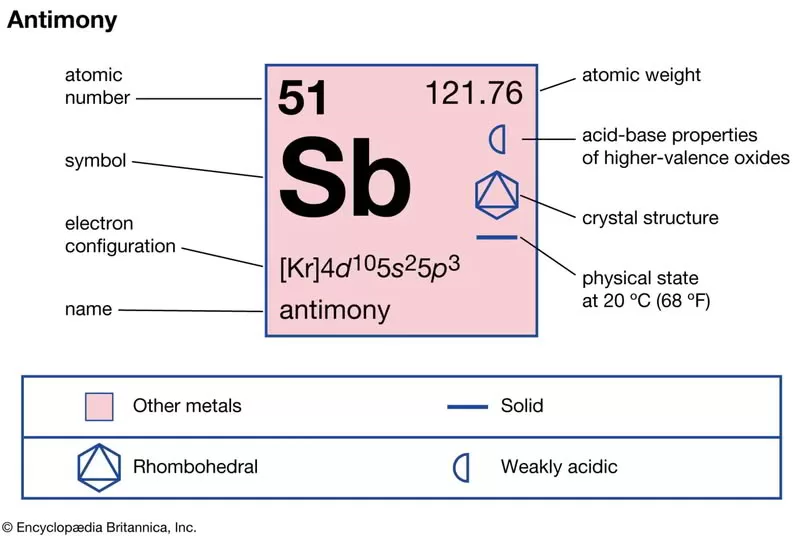
What is antimony used for in industry and technology?
Primary application sectors and market share
Antimony’s largest industrial role is as a precursor to antimony trioxide (Sb2O3), which is the dominant synergist in halogenated flame retardant systems for plastics, textiles and electrical components. Multiple industry analyses and government summaries indicate flame retardants account for roughly 40 to 55 percent of global antimony consumption, depending on the year and the report. (en.wikipedia.org)
The second major sector is metallurgical use, chiefly antimonial lead for lead‑acid battery grids and some bearing and solder alloys; many sources place batteries and lead‑alloys in the 20 to 35 percent range of total demand. Chemical and catalyst applications (including PET catalysts and specialty antimony compounds) plus glass, ceramics and pigments together represent the remainder, with semiconductors and niche electronics applications forming a small but growing slice of demand. Regional production remains concentrated, with China providing a very large share of primary supply and recycling contributing an important secondary stream for battery-derived antimony. (pubs.usgs.gov)
Typical functional roles in products
Antimony appears in products in three practical functional roles: as a chemical synergist and additive that enhances flame retardancy; as an alloying/hardening element that raises strength and creep resistance in lead and tin alloys; and as a chemical catalyst or stabilizer used in polymer manufacture and specialty chemical synthesis. In flame‑retarded polymer systems, Sb2O3 works together with halogenated flame retardants to form reactive antimony‑halogen species that interrupt combustion chemistry. In metallurgy, small percentages of antimony (typically a few percent) increase yield strength and hardness of battery grid alloys. As a catalyst, antimony species (notably Sb2O3) are used at low concentrations to accelerate polyester (PET) polymerization and to stabilize polymer processing. (en.wikipedia.org)

Antimony in plastics and flame retardant formulations
Role of antimony trioxide in polymers
Antimony trioxide (Sb2O3) is not a primary flame retardant but is the most widely used synergist for halogenated flame‑retardant systems. When a halogenated retardant decomposes under heat it releases hydrogen halides (for example HBr or HCl); Sb2O3 reacts to form volatile antimony halides that act in the gas phase to scavenge flame‑propagating radicals (H and OH), and it also promotes a more cohesive char in the condensed phase. That combined gas‑phase radical quenching plus condensed‑phase char promotion is why Sb2O3 remains standard in plastics such as PVC, ABS, polystyrene and many electrical housings where UL 94 or similar flammability ratings are required. Usage rates and particle grades are chosen to balance flame performance, color and dispersion; manufacturers sometimes partially replace Sb2O3 with inorganic or polymeric synergists to meet regulatory or formulation goals. (en.wikipedia.org)
Catalyst and stabilizer use in PET
Sb2O3 and other trivalent antimony compounds are the dominant catalysts for industrial PET polymerization because they provide high activity at moderate cost and give acceptable color and molecular‑weight control. Antimony catalysts are typically used at trace levels (parts per million of antimony in the final resin) and can influence reaction rate, intrinsic viscosity and byproduct formation such as acetaldehyde. Alternative catalysts based on titanium, germanium or aluminum exist and are used where lower antimony content or different optical/food‑contact properties are required, but antimony remains the commercial default for many bottle and fiber grades. Processing controls, scavengers and purification steps are used to minimize residuals and manage quality for sensitive applications. (ncbi.nlm.nih.gov)
Antimony in alloys and lead-acid batteries
Lead-alloys, battery grids and solders
Antimony is widely used as an alloying element in lead‑acid battery grids and in some solders because small additions (typically around 1 to 6 percent, depending on the alloy) markedly increase hardness, yield strength and castability compared with pure lead. In automotive and industrial battery grids, antimonial lead improves mechanical integrity during casting and service, helps control creep of the grid under compression, and can influence self‑discharge and cycle behavior; some historic formulations used higher Sb levels while modern formulations trend lower or substitute calcium/tin in parts of the battery to reduce maintenance. Antimony is also present in certain tin‑based solders to improve strength and thermal performance. (britannica.com)
Metallurgical benefits and recycling relevance
From a metallurgical perspective, antimony delivers hardness and improved corrosion resistance to lead alloys, which translates into longer grid life and reliable electrical contact in lead‑acid batteries. Because lead‑acid batteries are collected and recycled at high rates in many countries, secondary recovery is a major source of antimony feedstock: spent grids and other battery scrap are processed at secondary smelters to recover lead and grouped antimony as part of the recycled alloy stream. That recycling loop both supplies a significant share of industrial antimony used in alloys and buffers supply risk from concentrated primary mining. However, evolving battery alloy chemistry (for example wider adoption of low‑antimony or calcium alloys in some applications) and regulatory pressure on contaminants require smelters and recyclers to control composition and processing to meet modern specifications. (pubs.usgs.gov)
Antimony in electronics, semiconductors and infrared detectors
Dopant uses and compound semiconductors
Antimony is a common n‑type dopant in silicon and other semiconductor processes. Because Sb is a pentavalent element, small controlled additions substitute for silicon atoms and donate free electrons, producing n‑type conductivity with low diffusion rates and a relatively low segregation coefficient—properties that help produce stable, uniformly doped wafers for power devices, sensors and some photovoltaic applications. For deposition and epitaxy, antimony sources such as stibine or antimony fluxes are used in CVD and MBE processes to introduce controlled Sb concentrations.
Beyond elemental doping, antimony is a core constituent of III–V antimonide semiconductors (for example InSb, GaSb and their alloys). These antimonide compounds have narrow, tunable bandgaps, high carrier mobilities and strong infrared absorption, making them preferred materials for mid‑wave and long‑wave infrared photodetectors, high‑speed diodes and some high‑frequency transistors. Engineers select specific antimonide alloys to tune cutoff wavelength, responsivity and operating temperature for imaging and sensing systems. (See InSb and GaSb material families.)
Infrared detectors, diodes and thermoelectrics
Indium antimonide (InSb) and gallium antimonide (GaSb) remain standard for cooled mid‑infrared detectors used in thermal imaging, spectroscopy and certain defense sensors because of their high quantum efficiency and low noise at cryogenic temperatures. Antimonide heterostructures and strained‑layer superlattices allow wavelength engineering and improved detector performance, and research continues into monolithic integration of antimonide detectors on silicon to simplify readout integration.
Antimony‑containing compounds also appear in diode and thermoelectric technologies. Binary and ternary antimonides (including Sb‑rich chalcogenides such as Sb2Te3 and Bi-Sb alloys) exhibit favorable Seebeck coefficients and low thermal conductivity, so they are used in thermoelectric modules for cooling and power‑generation at specific temperature ranges. In short, antimony’s electronic and materials chemistry makes it a versatile ingredient across sensing, high‑frequency electronics and energy‑conversion devices.
Chemical forms of antimony and practical implications
Common compounds: oxides, sulfides and antimonates
Antimony occurs in several chemical forms that matter for performance and regulation. The oxides, especially antimony(III) oxide (Sb2O3) and antimony(V) oxide (Sb2O5), are the most commercially important: Sb2O3 is the primary flame‑retardant synergist and Sb2O5/antimonates appear in specialty retardant and catalyst roles. Antimony sulfide (Sb2S3), the mineral stibnite, is a common ore and is used historically as a pigment and today in niche semiconductor or photovoltaic research. Metal antimonates (for example sodium antimonate and complex mixed‑oxide antimonates) exist as higher‑valence, often more stable materials used where hydrolytic or thermal stability is required. These families differ in oxidation state, solubility and thermal behavior, which drives their selection for plastics, ceramics, catalysts and electronic materials. (en.wikipedia.org)
How chemical form affects performance and handling
Oxidation state and compound class determine both functional behavior and practical handling requirements. Trivalent Sb(III) oxides are reactive with halogen sources and provide gas‑phase radical quenching in flame‑retardant systems, while pentavalent antimonates are generally less volatile and can offer improved thermal or hydrolytic stability in some formulations. Sulfides and chalcogenides have distinct optical and electronic properties that make them attractive for photovoltaics and infrared devices but they require oxygen‑controlled processing to avoid oxidation. From a safety and regulatory standpoint, Sb(III) compounds are often more toxic and more tightly regulated than Sb(V) forms; dust control, respiratory protection and wastewater controls are common precautions when handling powders or processing scrap containing antimony. Selection therefore balances target performance (flame efficiency, catalytic activity, electronic properties) with manufacturability, end‑of‑life recycling and occupational/environmental controls. (atamanchemicals.com)
Supply chain, recycling options and regulatory risks for antimony
Major producing countries and supply concentration
Primary antimony supply is geographically concentrated. Recent commodity and government reports place China as the dominant producer and reserve holder, with a handful of other countries — including Tajikistan, Russia, Bolivia and Turkey — supplying most of the remainder. That concentration means changes to Chinese production or export policy can quickly tighten global markets and raise prices, and several recent public reports and market notices have flagged antimony as a supply‑risk or critical mineral for downstream industries. (pubs.usgs.gov)
Recycling from batteries and flame-retardant plastics
Secondary recovery is an important part of the antimony supply picture because antimony is used at appreciable levels in lead‑acid battery grids and in Sb2O3‑containing flame‑retardant plastics. High collection and recycling rates for lead‑acid batteries make spent batteries the most reliable secondary source of antimony in many countries; recovered antimony and antimonial lead from battery smelters supply alloy markets and reduce dependence on primary mining. Recycling antimony from flame‑retardant plastics is technically possible but commercially limited: low concentrations, mixed polymer streams and concerns about halogenated contaminants make economic recovery challenging without preconcentration or dedicated streams. (associationofbatteryrecyclers.com)
Key safety, disposal and regulatory flags
Regulatory and occupational controls focus on inhalation and environmental release of trivalent antimony species. International bodies and national agencies have evaluated antimony trioxide and related Sb(III) compounds for carcinogenic potential; IARC and other reviews have treated certain trivalent antimony forms as possibly or probably carcinogenic and regulatory classification has tightened in some jurisdictions. As a result, manufacturers and recyclers commonly apply dust controls, exposure monitoring, product stewardship limits for antimony residuals (for example in food‑contact PET) and end‑of‑life handling rules for Sb‑containing wastes. Separately, trade restrictions or export controls on critical minerals have been used as policy levers and can create near‑term supply disruption risk for industries that rely on antimony. Stakeholders should track evolving hazard classifications, workplace exposure limits and trade policy updates relevant to their supply chains. (iarc.who.int)